Steel is one of the founding materials of modern construction. As a material, its has high tensile strength, workability and cost-effectiveness. Steel has been found to be versatile in construction. It has been used to form the framework of skyscrapers, bridges, pipelines, and heavy machinery.

However, steel also has an inherent vulnerability: its susceptibility to corrosion. This is because of an electrochemical process, if left unmanaged, can degrade the material’s properties, compromise structural integrity, and lead to catastrophic failures. In this article, we are looking how to over this problem. We will look at the primary agents of corrosion, and the engineering methods for protecting it.
Why protect steel against corrosion?
The corrosion of steel is an electrochemical reaction wherein iron oxidizes in the presence of an electrolyte (typically water) and an oxidizing agent (typically oxygen) to form hydrated iron(III) oxide, commonly known as rust. This process is not merely cosmetic; it results in a loss of material cross-section, which directly reduces load-bearing capacity.
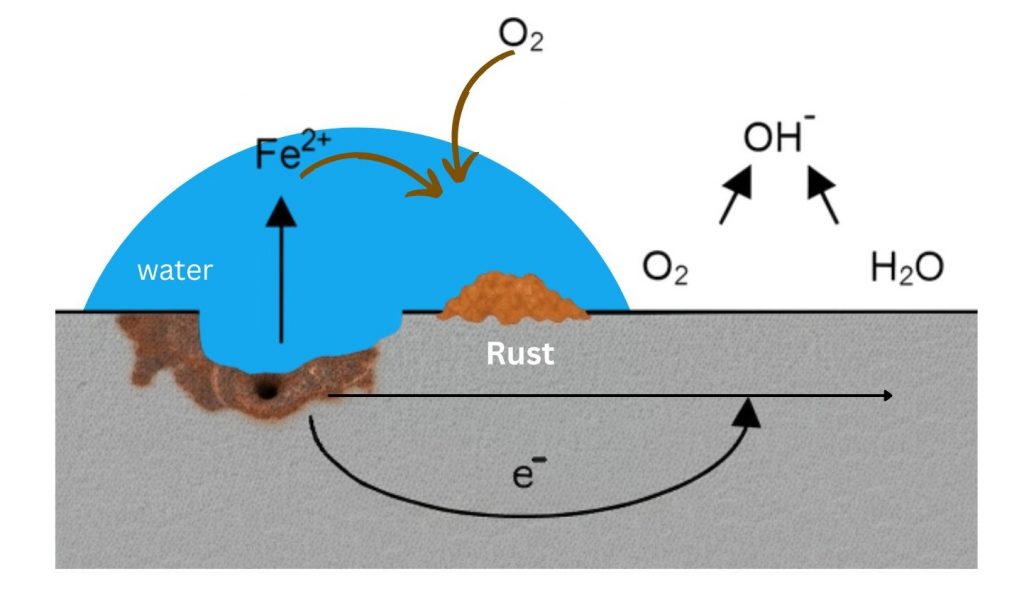
Effective corrosion mitigation is of engineering importance to help achieve the following when using steel for construction:
- To maintain structural integrity and ensure public safety: Preventing the degradation of critical components to ensure they perform to their designed specifications.
- To extend the service life of assets: By protecting the steel, the operational lifespan of infrastructure is increased which improves the return on investment.
- To reduce lifecycle costs: Proactively protect steel in structures can help minimize the significant costs associated with inspections, maintenance, repair and premature replacement.
- To comply with regulations and codes: Industry standards always require specific levels of corrosion protection for any type of steel structure.
What Causes Steel Corrosion? The Primary Culprits
The rate and type of steel corrosion are dictated by its environment. Key factors that accelerate corrosion include:
- Moisture and Humidity: Water is the electrolyte that enables the electrochemical reaction. High humidity and areas where moisture gets trapped are ideal for rust formation.
- Oxygen: As the primary oxidizing agent, the presence of oxygen is essential for the corrosion process to continue.
- Chlorides: Found in marine environments (sea salt) and deicing salts, chloride ions are extremely aggressive. They break down protective films on steel, leading to rapid and localized pitting corrosion.

- Industrial Pollutants: Atmospheric pollutants, primarily sulfur oxides (SOx) and nitrogen oxides (NOx), dissolve in atmospheric moisture to form acidic compounds like sulfuric and nitric acid. This lowers the pH of the electrolyte, significantly increasing the corrosion rate.
- Microbial-Induced Corrosion (MIC): Certain anaerobic and aerobic microorganisms can influence corrosion by altering the local chemistry at the steel’s surface, producing corrosive byproducts, or destroying protective films.
Proven Methods for Protecting Steel from Corrosion
There is a range of engineering techniques are used to protect steel. The best method depends on the environment, desired lifespan, budget, and maintenance access as shown below:
1. Barrier Coatings
This is the most common method of corrosion control, which involves isolating the steel substrate from the corrosive environment. Below are some of the barrier coating techniques used to protect steel:
- Industrial Paint Systems: These are typically multi-layer systems consisting of a primer for adhesion and inhibitive properties, an intermediate coat for building thickness, and a topcoat for UV and abrasion resistance. Formulations often include zinc-rich primers and epoxy or polyurethane topcoats.
- Epoxy Coatings: These thermosetting polymer coatings provide a hard, durable barrier with excellent adhesion and high resistance to chemicals, abrasion, and moisture. They are widely used in marine, offshore, and industrial process facilities.
- Powder Coating: A process where an electrostatically applied dry powder is cured under heat, fusing into a continuous, non-porous, and highly durable protective skin.

2. Galvanization
This process involves applying a coating of zinc to steel, typically through a hot-dip galvanizing process. The zinc provides protection in two ways: first as a physical barrier, and second as a sacrificial anode. If the coating is scratched, the more electrochemically active zinc will corrode preferentially, protecting the underlying steel in a process known as galvanic protection.

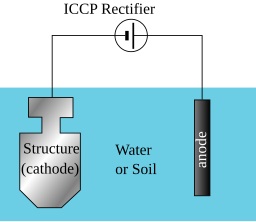
3. Cathodic Protection (CP)
CP is an electrochemical technique that mitigates corrosion by converting all of the active sites on the steel surface into cathodes. This is achieved by supplying an electrical current from an alternate source. There are two primary types:
- Sacrificial Anode Cathodic Protection (SACP): Involves electrically connecting a more active metal (a galvanic anode, such as zinc, aluminum, or magnesium) to the steel. The anode corrodes sacrificially, protecting the steel structure.

- Impressed Current Cathodic Protection (ICCP): Uses an external DC power source to impress a current through inert anodes (such as high-silicon cast iron or mixed metal oxide) onto the structure, forcing it to act as the cathode. This method is common for large-scale assets like pipelines and marine structures.
4. Corrosion Inhibitors
These are chemical substances added in small amounts to an environment to decrease the corrosion rate. They work by adsorbing onto the metal surface to form a protective film. Inhibitors are most effective in closed-loop systems like boilers, cooling water systems, or for temporary protection during shipping.
6. Design for Corrosion Control
Proactive design is one of the most effective and low-cost measures. Key principles include:
- Avoid trapping moisture: Design to eliminate crevices and low points where water can collect.
- Ensure proper drainage: Include weep holes and drainage paths so water can escape freely.
- Design for maintenance: Ensure all parts of a structure are accessible for future inspection, cleaning, and re-coating.
Conclusion
While the corrosion of steel is a natural process, it is entirely manageable through the diligent application of sound engineering principles. By understanding what causes rust and strategically implementing mitigation methods from advanced coatings and cathodic protection to smart material selection and design, we can ensure the long-term integrity, safety, and economic viability ofcritical steel infrastructure.
Please don’t forget to subscribe to our newsletter!
YOU MAY ALSO LIKE TO READ
- A Simple Guide to Runoff ManagementWhether you are working on a small infrastructure project or a large one, rainwater (runoff) is always going to be an issue. That’s why runoff management is a critical part of sustainable land development and environmental protection. As more parts of the world become urbanized and climate patterns continue to… Read more: A Simple Guide to Runoff Management
- A Billion Dollar Problem: What Construction Waste Management Means for the IndustryIt is estimated that nearly 30% of the world’s solid waste comes from construction and demolition (C&D) activities. This includes everything from broken concrete, wood, metal, drywall, bricks, glass, plastics, asphalt, to packaging materials. Most of this waste ends up in landfills, contributing to environmental degradation and increasing costs for… Read more: A Billion Dollar Problem: What Construction Waste Management Means for the Industry
- 5000 Years of Computing: Our Engineering Journey to Quantum ComputingFor over a millennium, human ingenuity has been entwined with the development of tools to aid computation. From the use of the abacus beads to the barely comprehensible entanglement of quantum bits, computing has evolved not only in speed and complexity but also in the way engineers use it to… Read more: 5000 Years of Computing: Our Engineering Journey to Quantum Computing

